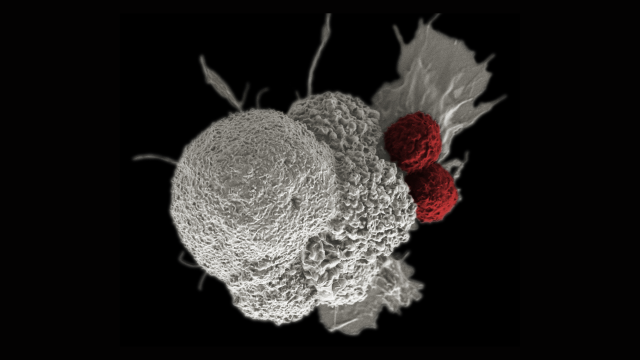
In a landmark study, a patient’s advanced breast cancer regressed completely after treatment with immune cells that recognized products of mutations specific to her cancer.
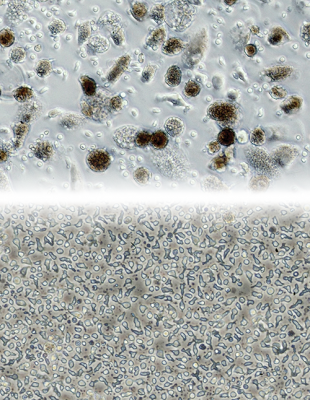
At the top of the image are cells isolated from a patient’s tumor directly after it was removed by surgery. The sample includes a combination of dark tumor cells, tumor-infiltrating lymphocytes (TILs) and other inflammatory cells. Below, the same culture is shown after two to three weeks of growth. This is a pure sample of TILs and is used to select lymphocytes that are able to identify the products of mutations unique to the patient’s tumor. Once multiplied, TILs are then infused into the patient.
Credit: James Yang, CCR, NCI, NIH
In an ongoing NCI-led clinical trial at the NIH Clinical Center, a patient with metastatic breast cancer has experienced complete regression of her disease after treatment with an experimental immunotherapy developed in CCR. Despite multiple prior cancer treatments, her cancer continued to enlarge and spread until she received this new treatment. The case report, published in Nature Medicine, highlights the potential of immunotherapy for difficult-to-treat solid tumors.
Although cancer immunotherapies have brought about complete regression of disease in some patients, certain types of cancer, including breast cancer, have been difficult to treat with these approaches. The experimental approach being tested in the current trial, developed by CCR Surgery Branch Chief Steven A. Rosenberg, M.D., Ph.D., and colleagues, uses immune cells selected for their ability to recognize cells with mutations specific to a patient’s tumor to fight their disease—a strategy that Rosenberg says may serve as a blueprint to treat many cancers.
This type of immunotherapy, called adoptive cell transfer (ACT), uses cancer-fighting cells from a patient’s own immune system that are grown in large numbers in the laboratory and then infused into the patient. The specific form of ACT being investigated uses immune cells that have been isolated directly from a patient’s tumor, known as tumor-infiltrating lymphocytes (TILs). The key to this approach is the identification and use of only TILs that specifically recognize proteins encoded by genetic mutations present in the patient’s cancer.
The TILs used to successfully treat this patient’s breast cancer were selected after researchers compared the DNA and RNA from one of her tumors to her normal tissue and identified 62 mutations unique to her cancer. Next, the TILs isolated from her tumor were tested for their ability to recognize the products of these mutations. TILs recognizing proteins encoded by four different cancer-specific mutations were then selected for treatment. Along with the tumor-specific immune cells, the patient was given pembrolizumab, an immune system checkpoint inhibitor that allows cancer-fighting cells to remain active in the microenvironment of a tumor, and interleukin-2, which promotes immune cell growth.
More than three years after the treatment, the patient’s breast cancer has not returned. The team has also seen this type of immunotherapy trigger tumor regression in patients with liver cancer, colorectal cancer and cervical cancer, offering hope that it might be a broadly effective treatment strategy.