News and Events
New Strategy Shows Promise Against Deadly Breast Cancer in the Brain
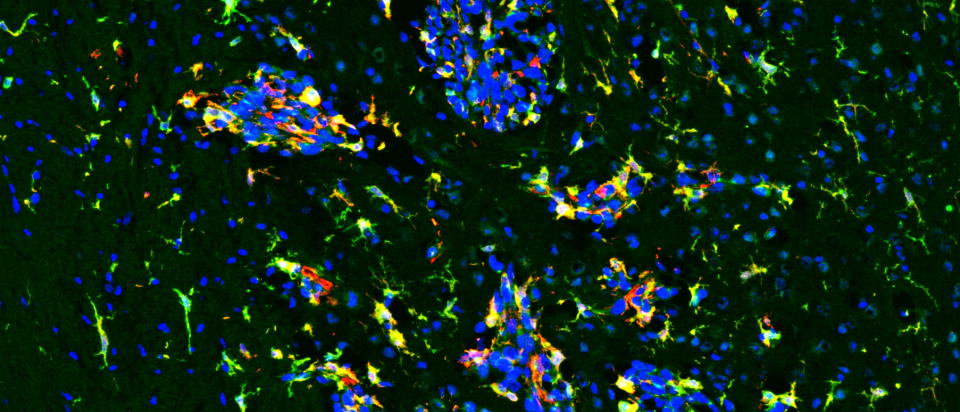
A new NIH study points to a promising strategy for treating aggressive breast cancer that spreads to the brain, a complication with few effective options. Learn how blocking a key brain cell survival pathway could open the door to future therapies.
Read MoreEmpowering others to contribute to rare neuroendocrine tumor research
Lacey Koelling was diagnosed with paraganglioma tumors in 2018, which began to metastasize five years later. She enrolled in an NIH trial to contribute to research on this rare cancer.
Read MoreAn observant patient opens new doors for cancer screening
David Drash enrolled in a CCR clinical trial when he was diagnosed with BAP1 tumor predisposition syndrome (TPDS) in 2022. He noticed that everyone in his family with BAP1 TPDS had white lines on their fingernails; his observation led to the discovery of a new detection tool.
Read MoreCelebrating CCR Careers: Alan Rein, Ph.D.
Alan Rein, Ph.D., is a known expert in the field of viral assembly, particularly in retroviruses like the human immunodeficiency virus (HIV). After an extensive career studying molecular mechanisms of retroviral replication and pathogenesis, he has announced his retirement.
Read MoreClinical trial researching MRI software for mapping prostate cancer
A clinical trial led by Ismail Baris Turkbey, M.D., Senior Clinician in the Molecular Imaging Branch, is researching a software that can analyze radiographic images of the prostate for cancer detection and diagnosis.
Read MoreMutant RAS can shift other proteins’ positions inside cancer cells
Mutant RAS proteins help shuttle other proteins out of the cell nucleus and into the cytoplasm, where some can promote cancerous growth.
Read MoreClinical trial researching combination drug therapy for Ewing sarcoma and rhabdomyosarcoma
A clinical trial led by Christine M. Heske, M.D., Investigator in the Pediatric Oncology Branch, is researching combination drug therapy for adolescents and young adults with Ewing sarcoma and rhabdomyosarcoma.
Read MoreClinical trial researching drug therapy for adults with colorectal or gastroesophageal cancer
A clinical trial led by Nicholas D. Klemen, M.D., Physician-Scientist Early Investigator in the Surgery Branch, is researching drug therapy for adults with colorectal or gastroesophageal cancer.
Read MoreClinical trial researching drug therapy for adults with HIV-related diseases
A clinical trial led by Ramya Ramaswami, M.B.B.S., M.P.H., Lasker Clinical Research Scholar in the HIV and AIDS Malignancy Branch, is researching drug therapy for two HIV-related diseases.
Read MoreClinical trial researching CAR T-cell therapy for leukemia and lymphoma
A clinical trial led by Jennifer N. Brudno, M.D., Associate Research Physician in the Surgery Branch, is researching CAR T-cell therapy for chronic lymphocytic leukemia and small lymphocytic lymphoma.
Read MoreClinical trial researching combination drug therapy for genitourinary tumors
A clinical trial led by Andrea B. Apolo, M.D., Senior Investigator in the Genitourinary Malignancies Branch, is researching combination drug therapy for rare genitourinary tract tumors.
Read More