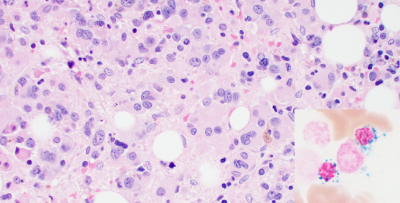
The NIH Myeloid Malignancies Clinical Program focuses on the rapid translation of science to novel, first-in-disease clinical trials with the aim of developing new science-driven therapy approaches in adult and pediatric patients with myeloid malignancies, especially MDS and secondary AML.
Myeloid Malignancies Program
A trans-NIH multidisciplinary network of researchers and clinicians dedicated to improving early detection, diagnosis, prognosis and development of novel therapeutics for myeloid malignancies.
Recent Publications
View a listing of our most recent publications.
Myeloid Malignancies Program
About
Our Vision
The vision of the NIH Myeloid Malignancies Program is to successfully control, cure and prevent MDS and AML.
The mission of the trans-NIH Myeloid Malignancies Program (MMP) is to develop a comprehensive program aimed at understanding, diagnosing, and treating the heterogeneous collection of myeloid malignancies that arise in children and adults, especially the Myelodysplastic Syndromes (MDS) and secondary or therapy-related Acute Myelogenous Leukemia (AML). Our objective is to establish a trans-NIH clinical research program at the NIH Clinical Center to allow rapid translation of intramural science and the development of novel therapies for patients with MDS and AML.
This program functions as a clinical scientific unit with regular protocol and research progress meetings. It also serves as a focal point for fellows and medical staff education in the practice of translational clinical research in the myeloid malignancies. We aim to utilize new intramural opportunities and initiatives to stimulate translational research in MDS and AML.
There are more than 10,000 new cases of MDS diagnosed in the U.S. every year. These blood cancers remain difficult to treat due to vast differences in the patient population including great genetic diversity, complicated clinical presentation with similarities to several overlap syndromes, and lack of highly effective and long-lasting treatments. These difficulties inspired the trans-NIH MMP to consolidate expertise and create an interactive and collaborative environment to foster myeloid malignancy care and research through innovative laboratory studies, state-of-the-art research, and promising clinical applications. The overarching goal is to develop novel therapeutic targeting strategies that may significantly alter the natural history of these disorders.
Join Our Email List
To join the Myeloid Malignancy Interest Group email list, please visit the Myeloid Malignancy Interest Group Listserv homepage, then click the “Subscribe or Unsubscribe” link in the right sidebar.
Clinical Trials
Have questions about Myeloid Malignancies Program clinical trials?
Please contact Rebecca Alexander via email.
rebecca.alexander@nih.gov
Team
The Myeloid Malignancies Program consists of physician-scientists and investigators from multiple institutes within the National Institutes of Health (NIH), including the National Cancer Institutes (NCI), the National Heart, Lung, and Blood Institute (NHLBI), and others. If you are interested in participating in this trans-NIH program, please contact a member of our leadership team. View a list of participating NIH investigators.
Clinical Director
Translational Director
Steering Committee
Our Research Program
We are developing a comprehensive program aimed at understanding and treating myeloid malignancies in children and adults. Our research program has four overarching themes:
- Pre-clinical models to study MDS biology and therapy
- Post-transcriptional regulation in MDS/AML
- Germline predispositions to myeloid malignancies
- Role of the immune system in control of MDS/AML
Join Our Email List
To join the Myeloid Malignancy Interest Group email list, please visit the Myeloid Malignancy Interest Group Listserv homepage, then click the “Subscribe or Unsubscribe” link in the right sidebar.