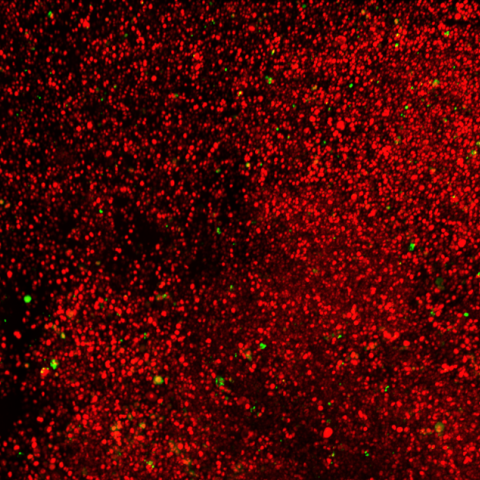
Florescent microscopy showing a layer of cancer cells (red) with rare cancer stem cells (green) in a primary tumor from the human breast cancer cell line.
Image courtesy of Dr. Lalage Wakefield.
A new finding suggests why breast cancer cells are typically more difficult to treat after they spread beyond their primary tumor site. Lalage M. Wakefield, D.Phil., senior investigator in the Laboratory of Cancer Biology and Genetics co-led the study with Maja H. Oktay, M.D., and John Condeelis, Ph.D., Albert Einstein College of Medicine. As reported Dec. 15, 2021, in Nature Communications, when cancer cells pass through a specialized portal to enter the blood stream, they come in contact with macrophages (a type of white blood cell), which reprogram cancer cells into more aggressive, therapy-resistant, stem-like cells.
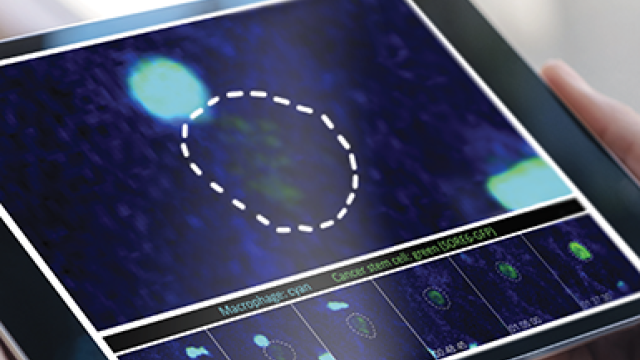
To study intact breast cancer tumor and tumor-supporting cells, the researchers used a powerful time-lapse imaging technique in living mice, using fluorescently labeled breast cancer stem cells that were implanted into the mice. This cutting-edge technology enabled the team to observe individual breast cancer cell behavior over time in the cell’s native habitat. Their first observation: cancer stem cells were enriched near macrophages — particularly macrophages found in specialized portals into the blood stream.
In a process Wakefield dubbed the “macrophage kiss,” contact between macrophages and non-stem cancer cells induced a stem-like character, mediated by a cellular process known as Notch-Jagged signaling. “A consequence of this is that tumor cells entering circulation have a massive enrichment in stem-like cells, presumably as a result of the tumor cells squeezing past macrophages to enter the bloodstream,” Wakefield said. These stem-like cells represented only one percent of cancer cells in the primary tumor but as much as 70 percent of cancer cells in the blood stream.
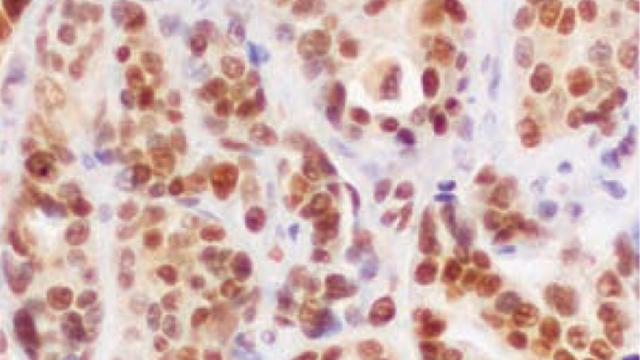
Looking at breast cancer tumor samples from patients, the researchers found a correlation between the number of macrophage-containing portal structures and the number of cancer cells with stem-like characteristics. This finding in human tissue further supports the idea that macrophage contact coaxes a non-stem cancer cell into a stem-like cell.
The researchers next step is to understand the detailed mechanisms of cancer cell reprogramming following macrophage contact. “If we understand it better, we can disrupt and reduce metastatic spread,” Wakefield said. She’d also like to know if similar mechanisms operate at the metastatic site and if other cell types in the metastatic environment, other than macrophages, can reprogram cancer cells too.
While this work focused on breast cancer, these macrophage-containing portal structures are found in multiple cancer types. Wakefield suspects this work is relevant for other solid tumors, as stem-like tumor cells are found in many other cancers.