Lichun Ma, Ph.D.
- Center for Cancer Research
- National Cancer Institute
- Building 41, Room A100B
- Bethesda, MD 20892
- 240-760-6085
- lichun.ma@nih.gov
RESEARCH SUMMARY
Lichun Ma, Ph.D., is a systems biologist with a specific focus on understanding the intrinsic tumor biology of liver cancer using single-cell and spatial approaches. Her research centers on tumor heterogeneity in the context of tumor initiation and evolution with the goal of improving early detection and therapeutics for liver cancer. In addition to her role as a principal investigator within the Cancer Data Science Laboratory, Dr. Ma is also affiliated with the CCR Liver Cancer Program.
Areas of Expertise

Lichun Ma, Ph.D.
Research
Tumor heterogeneity is a major contributor to therapeutic failures and lethal outcomes in liver cancer. A better understanding of tumor heterogeneity is critical for improving liver cancer treatment. Using cutting-edge technology in single-cell and spatial omics assays, my research program will develop novel approaches to understanding tumor heterogeneity in the context of tumor initiation and evolution. Our long-term goal is to improve early detection and therapeutics for liver cancer.
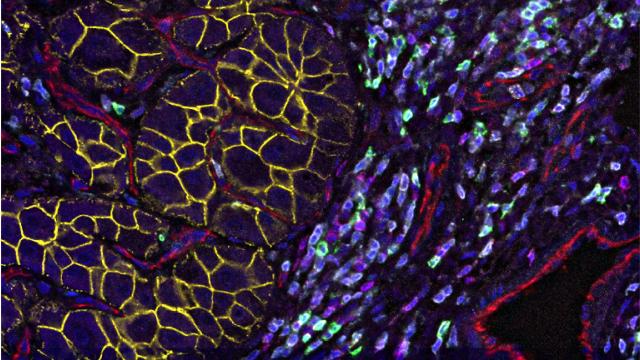
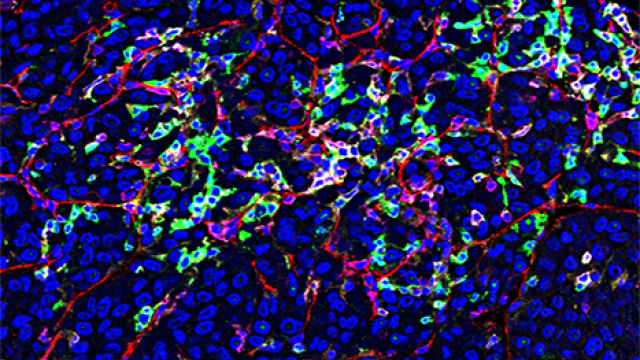
1) Tumor heterogeneity. Tumor heterogeneity is a common feature in liver cancer. However, what determines the observed different degrees of cellular heterogeneity among tumors is not known. One possible mechanism is that distinct cells within each tumor serve as cells of origin in tumorigenesis among patients. Thus, we are interested in understanding the tumor cells of origin. In addition, the tumor microenvironment (TME) has been recognized as a key player in promoting tumor heterogeneity. Within a tumor ecosystem, tumor cells continuously communicate with the TME through cell adhesion, signaling molecules and small vesicles. Our ability to dissect the communication networks connecting the tumor with the TME would allow a better understanding of tumor heterogeneity. Moreover, cellular functions are closely related to the cell’s location within the tumor. Deciphering the spatial information of the tumor and stromal/immune cells would provide a critical framework in which to understand their communications.
2) Tumor evolution in response to treatment. Tumor evolution is a key feature of tumorigenesis and plays a pivotal role in driving intratumor heterogeneity, treatment failure, escape from immune surveillance and ultimately, patient prognosis. We have demonstrated that a tumor’s evolutionary trajectory may change over time in response to immunotherapy based on single-cell profiling of core needle tumor biopsies from liver cancer patients before and after treatment. It is known that only ~20% of liver cancer patients respond to immunotherapy, and it is unclear why some patients respond to immunotherapy while others do not. To improve liver cancer treatment, we use longitudinal clinical tumor specimens and animal models to identify molecular features that are responsible for tumor evolution.
Publications
- Bibliography Link
- View Dr. Ma's Google Scholar Bibliiography.
Biography

Lichun Ma, Ph.D.
Dr. Ma received her Ph.D. degree in Electronic Engineering at the City University of Hong Kong in 2016. After a one-year postdoctoral fellowship at Nanyang Technological University, Singapore, she joined NCI in 2017 as a postdoctoral fellow where she studied cancer biology using single-cell techniques. She initiated her independent research program at NCI as a Stadtman Investigator in 2022. Dr. Ma has a strong background in mathematics, information theory and machine learning. She received many awards during her training, including the NCI CCR Excellence in Postdoctoral Research Transition award. Her recent work on tumor heterogeneity and microenvironmental reprogramming in liver cancer was showcased in the 2019-2020 NCI Center for Cancer Research Milestones publication.
Job Vacancies
We have no open positions in our group at this time, please check back later.
To see all available positions at CCR, take a look at our Careers page. You can also subscribe to receive CCR's latest job and training opportunities in your inbox.
News
Team
Covers

Dissecting Tumor Heterogeneity by A Single-cell Approach
On the cover: Liver cancers are molecularly and biologically heterogenous, which contributes to their therapeutic failures and lethal outcomes. Ma et al. (pp. 418–430) have developed a single-cell-based approach to determine tumor cell communities (depicted as dice emptying out of a jar) and uncover a unique liver cancer ecosystem relevant to immune therapy. A tumor cell community is depicted by dice with different colors and shapes, where color represents cell type and shape represents unique molecular properties. Artwork by Ethan Tyler.
Ma L, Hernandez MO, Zhao Y, Mehta M, Tran B, Kelly M, Rae Z, Hernandez JM, Davis JL, Martin SP, Kleiner DE, Hewitt SM, Ylaya K, Wood BJ, Greten TF, Wang XW. Tumor cell biodiversity drives microenvironmental reprogramming in liver cancer. Cancer Cell 36: 418-30, 2019; PMID: 31588021.
Resources
Single-cell Atlas in Liver Cancer (scAtlasLC)
scAtlasLC is a publicly available data portal of single-cell transcriptomic profiles of tumor cell communities in hepatocellular carcinoma and intrahepatic cholangiocarcinoma. scAtlasLC can be used to evaluate gene expression in malignant cells and various non-malignant cells in liver cancer. It can be further used to determine gene expression in different subtypes of stromal cells and immune cells.