Eros Lazzerini Denchi, Ph.D.
- Center for Cancer Research
- National Cancer Institute
- Building 37, Room 2144B
- Bethesda, MD 20892
- 240-760-7031
- eros.lazzerinidenchi@nih.gov
RESEARCH SUMMARY
A key feature of a living organism is to preserve its genome integrity. To this end, cells use specialized proteins that survey the genome to detect and repair any lesion in the DNA. These vital processes, however, must be kept in check at naturally occurring DNA breaks; the ends of linear chromosomes. For this reason, all eukaryotic cells have telomeres, essential nucleoprotein structures that cap chromosome ends. Perturbation of telomere function frequently occurs in human pathologies such as cancer and aging. In these pathologies, telomere DNA is progressively lost, and a DNA damage response is initiated at chromosome ends. Our lab focuses on two fundamental aspects of telomere biology: the mechanism by which functional telomeres prevent DNA damage activation and the in vivo consequences of telomere dysfunction.
Areas of Expertise

Eros Lazzerini Denchi, Ph.D.
Research
We study the mechanisms by which telomeres protect chromosome ends, thus ensuring chromosome and genome stability. Our lab focuses on the identification of telomere-associated proteins to define their role in preventing activation of the DNA damage response pathway and suppression of end-to-end chromosomal fusions. We also study how telomere-associated proteins regulate the balance between telomere elongation and telomere erosion, thus controlling the replicative potential of normal as well as cancer cells. Identification of novel factors that play a role in telomere biology is carried out in the lab using proteomics as well as genetic screens.
A complementary study in our lab is focused on the in vivo consequences of telomere dysfunction. Telomere length homeostasis plays a critical role in cellular and organism survival. However, in humans, this process is inefficient and progressive telomere shortening is observed in tissues with a high cellular turnover. In the lab, we probe the role of telomere shortening in aging and tumor onset using mouse models that recapitulate telomere dysfunction in stem cells and other cellular compartments.
The Lazzerini Denchi Lab, March 2022
Publications
Identification of modulators of the ALT pathway through a native FISH-based optical screen
Native FISH: A low- and high-throughput assay to analyze the alternative lengthening of telomere (ALT) pathway
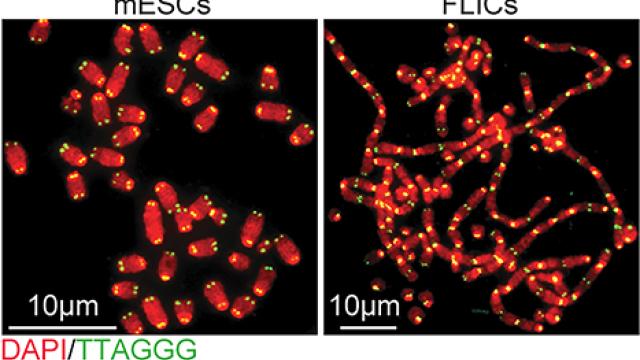
TRF2-mediated telomere protection is dispensable in pluripotent stem cells.
TZAP: a telomere-associated protein involved in telomere length control
Different requirement of functional telomeres in neural stem cells and terminally differentiated neurons
Biography

Eros Lazzerini Denchi, Ph.D.
Dr. Eros Lazzerini Denchi earned his M.S. in biology from the Universita’ Statale di Milano and received his Ph.D. in molecular oncology from the European Institute of Oncology (Milan, Italy) working in the laboratory of Prof. Kristian Helin. He completed his postdoctoral training in the laboratory of Prof. Titia de Lange at The Rockefeller University, New York, and worked as an independent researcher at The Scripps Research Institute, La Jolla, CA. Dr. Lazzerini Denchi became a Senior Investigator in 2021.
Job Vacancies
We have no open positions in our group at this time, please check back later.
To see all available positions at CCR, take a look at our Careers page. You can also subscribe to receive CCR's latest job and training opportunities in your inbox.