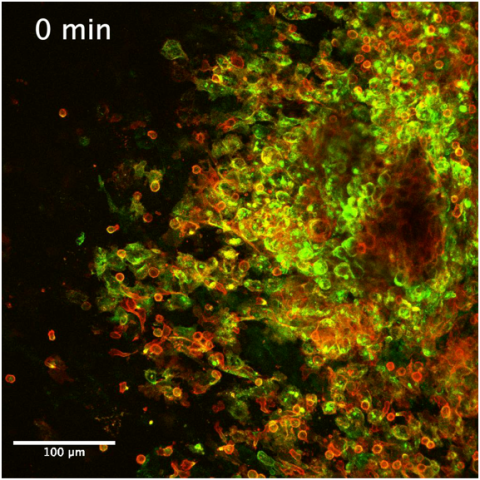
Live imaging screenshot of colorectal peritoneal metastasis.
Adults with peritoneal carcinomatosis may be eligible to participate in a clinical trial at the NIH Clinical Center.
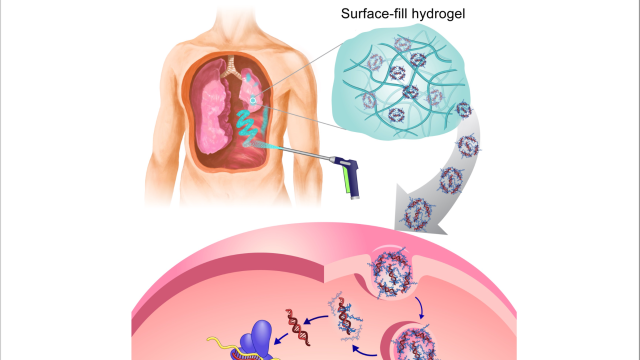
Andrew M. Blakely, M.D., Assistant Research Physician in the Surgical Oncology Program, is leading a study of surgical treatment for adults with peritoneal carcinomatosis. When cancers of the gastrointestinal tract or female reproductive organs spread, they can cause tumors to grow in the peritoneum, a thin layer of tissue that lines the abdomen and covers the abdominal organs. This type of cancer often responds well to surgery that removes areas involved with disease, called cytoreduction, in conjunction with a procedure called HIPEC (heated intraperitoneal chemotherapy). After a surgeon removes as many cancer cells as possible from the abdominal cavity, the cavity is filled with heated chemotherapy drugs. The purpose of HIPEC is to target microscopic areas of disease left behind by surgery.
In this trial, a tissue-testing platform in the lab will be used to study patients’ tumor tissue. The patient will undergo cytoreduction and HIPEC. During the surgery, some tumor tissue will be taken to the lab and exposed to two equivalent HIPEC treatment regimens, simulated in the tissue-testing platform. A small amount of tumor tissue will be exposed to the HIPEC treatment in the operating room, immediately removed, and placed on the tissue-testing platform. The tumor tissue exposed to simulated versus actual HIPEC will be compared to evaluate the ability of the tissue-testing platform to mirror what is happening in the operating room. The goal of this study is to validate the tissue-testing platform, which will bring researchers one step closer to providing individualized HIPEC treatment for future patients with peritoneal carcinomatosis.
Clinicaltrials.gov identifier: NCT04847063
NCI Protocol ID: NCI-21-C-0012
Official Title: Individualized Response Assessment to Heated Intraperitoneal Chemotherapy (HIPEC) for the Treatment of Peritoneal Carcinomatosis From Ovarian, Colorectal, Appendiceal, or Peritoneal Mesothelioma Histologies
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.