In the Spotlight
Former Pediatric Oncology Branch patient donates toys to NIH Pediatric Clinic
In December, former patient Julie Jones donated over 1,000 toys to the Pediatric Clinic at the NIH Clinical Center. Jones is a former patient in the Pediatric Oncology Branch who was treated more than 20 years ago for alveolar rhabdomyosarcoma. She says, “I remember what it was like. What these children are going through at such a young age really breaks your heart. I’ve often said there’s nothing greater than seeing the smile on the face of a sick child.”
Read MoreNeurofibromatosis type 1 patient attends Camp Fantastic for the first time
Thirteen-year-old Dom was diagnosed with neurofibromatosis type 1 at birth. Because of Dom’s diagnosis, he hasn’t always had the opportunity to do the things the average teenager does, but his first year at Camp Fantastic changed that. Camp Fantastic, an annual week-long camp for pediatric cancer patients and survivors, gave Dom the opportunity to try lots of new things and some familiar ones in a setting of support, safety and friendship.
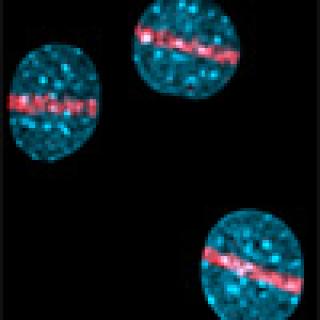
Read MoreLandmark discovery: H2AX as a sensor of DNA damage
William Bonner’s curiosity about histones led to the discovery that the protein H2AX is altered in response to harmful DNA damage called a double-strand break. This powerful tool has led to breakthroughs in both basic and clinical cancer research.
Read MoreFDA grants orphan drug status to selumetinib for neurofibromatosis type 1 (NF1) treatment
The U.S. Food and Drug Administration granted orphan drug status in February to selumetinib for use in patients with the genetic disorder neurofibromatosis type 1 (NF1), who often develop tumors of the peripheral nervous system. Receiving orphan drug designation is a helpful step for selumetinib.
Read More