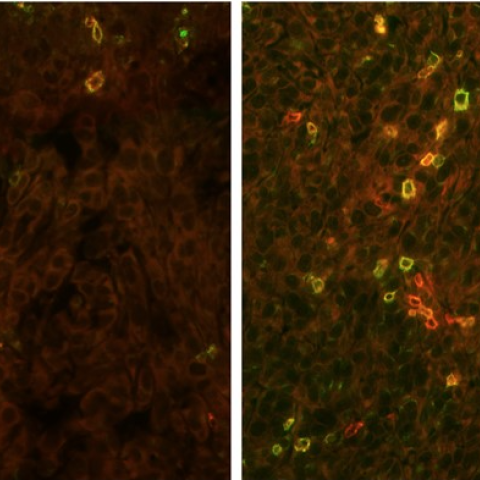
New research shows that mizoribine, which inhibits the production of purines, can increase the ability of immune cells to target cancer cells following immunotherapy (right panel). In contrast, fewer immune cells are able to penetrate tumors treated with immunotherapy without mizoribine (left panel).
Image credit: Ruppin, et al.
New research in mice and the lab suggests that blocking purine synthesis in high-ASS1 expressing tumors boosts the effectiveness of immunotherapy. The results were published August 31, 2020, in Nature Cancer.
Ayelet Erez, M.D., Ph.D., at the Weizmann Institute in Israel, and Eytan Ruppin, M.D., Ph.D., of CCR’s Cancer Data Science Laboratory, first started studying the functional role of the enzyme ASS1 in cancer seven years ago. They found that ASS1 influenced the composition of the building block molecules that cancer cells use to create DNA and RNA. They found that low ASS1 expression supports cancer proliferation by increasing pyrimidines to create more genetic material, and this switch to pyrimidines occurring in these tumors made them more susceptible to treatment with immunotherapy drugs.
In their more recent work published now, the Ruppin lab analyzed data derived from human samples in The Cancer Genome Atlas (TCGA), and found that some lung, colon and breast cancer tumors expressed an abundance of ASS1. Curiously, the tumors with high levels of ASS1 were more likely to have spread to other parts of the body and responded less to immunotherapy.
“Since high pyrimidine increases response to immunotherapy, we hypothesized that these tumors may actually have increased purine metabolism,” says Ayelet Erez. “We set out to investigate if indeed this is the case, and if so, we asked if we can reverse the increased purine production and increase pyrimidine levels and subsequently increase the response to immunotherapy of high ASS1 tumors.”
Through a series of experiments in breast and lung cancer cells performed in the Erez lab, the researchers confirmed that ASS1 increases the production of purines and uncovered the metabolic alterations underlying this phenomenon. Next, in mice induced to grow breast cancer tumors, they explored the effects of a drug that blocks purine production, called mizoribine, finding that this drug restricted tumor growth and made the tumors, which would typically be resistant to immunotherapy, responsive to the treatment. Furthermore, following treatment with mizoribine, tumors and T cells taken from lung cancer patients that initially did not respond to immunotherapy, demonstrated a significant response to immunotherapy.
“We are now in the midst of experimentally confirming the potential beneficial effects of additional FDA-approved small molecules that we’ve predicted to make high ASS1-expressing tumors even more responsive to immunotherapy,” says Ruppin. “Our preliminary preclinical results, led by the Erez lab, are very encouraging.”


