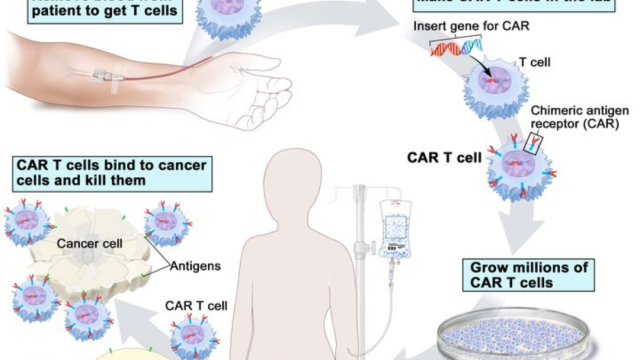
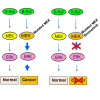
Normally BRAF (green) stimulates (phosphorylates) MEK (yellow), and in turn, MEK stimulates ERK (pink), leading to normal cell proliferation. Mutated BRAF, carrying the V600E mutation (red), overstimulates MEK, which overstimulates ERK. This leads to increased cell proliferation. In pigmented skin cells called melanocytes, this leads to melanoma. In B-cells, this leads to hairy cell leukemia (HCL). Inhibition of BRAF V600E by encorafenib blocks the overstimulation of MEK by BRAF V600E. The BRAF inhibitor may also increase stimulation through normal BRAF, leading to rashes and fever. Binimetinib blocks the overstimulation of ERK by MEK in both normal and HCL cells. This can lead to increased killing of HCL and decreased toxicity in normal cells.
Image credit: Robert Kreitman, M.D.
Adults with BRAF-mutant hairy cell leukemia, that did not respond to or came back after treatment, may be eligible to participate in a clinical trial at the NIH Clinical Center.
Robert J. Kreitman, M.D., Senior Investigator in the Laboratory of Molecular Biology is leading a study of a treatment for hairy cell leukemia (HCL). HCL is a rare, slow-growing cancer of the blood in which a person’s bone marrow makes too many B cells, a type of white blood cell that fights infection. These extra B cells are abnormal and look “hairy” under a microscope. Chemotherapy helps most cases of HCL but not for long. Most people with HCL have a BRAF gene mutation that can increase the growth of cancer cells; both BRAF and MEK send chemical signals that help to drive the growth and spread of cancer cells. Encorafenib is a small molecule that targets and inhibits BRAF signaling, while binimetinib, also a small molecule, targets and inhibits MEK signaling. Researchers want to see if this drug combination of encorafenib and binimetinib has an improved response rate than other drugs previously used to treat HCL.
Clinicaltrials.gov identifier: NCT04324112
NCI Protocol ID: NCI-20-C-0076
Official Title: Phase 2 Trial of Encorafenib Plus Binimetinib for Patients With BRAF V600E Mutated Relapsed/Refractory HCL
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.