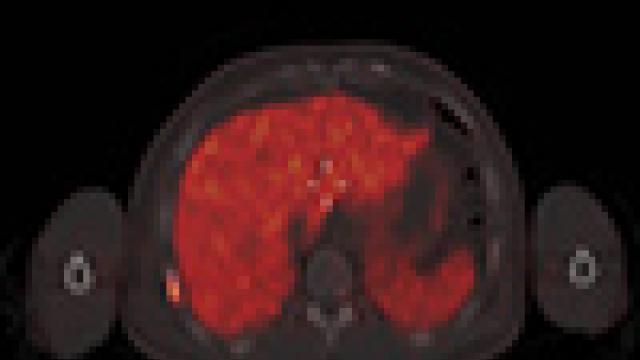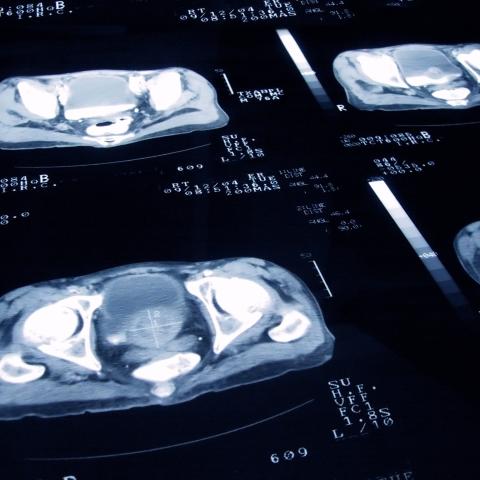
A series of images depicting computed tomography (CT) scans of prostate cancer.
Image Credit: iStock
Hormone therapy for prostate cancer, one of the most common cancers in men, can cause side effects so significant that some patients forgo treatment or end treatment prematurely, compromising their care. Researchers, led by Deborah E. Citrin, M.D., Senior Investigator in the Radiation Oncology Branch, are now investigating the benefits of delivering hormones directly into the tumor, with the goal of improving the quality of life for patients undergoing this type of prostate cancer treatment.
Citrin’s team is seeking patients for a phase I clinical trial to test the tolerability and effectiveness of Biolen™, a medication-secreting implant for localized prostate cancer. Measuring about one millimeter by 15 millimeters in size (like a couple of grains of rice end to end), the Biolen implant is a tiny device that locally releases a drug called bicalutamide. This drug halts prostate cancer cell growth by blocking the hormone testosterone. The team will combine Biolen implants with localized radiation therapy to shrink prostate cancer tumors.
While some patients with localized, slow-growing tumors can choose to watch and wait, those with intermediate or high-risk prostate cancer do need some form of treatment. For many patients, radiation combined with hormone therapy is the best option, but hormone therapy can span months to years depending on the characteristics of a patient’s tumor. Common side effects of hormone therapy include loss of libido, severe hot flashes, weight gain, mood changes, muscle loss, breast tenderness and altered metabolism. “Reducing the burden of side effects for patients is one of my primary goals,” says Citrin, who wants to avoid patients quitting treatment due to the side effects.
In this trial, Citrin and her team are using magnetic resonance imaging (MRI) to determine if and how they can inject Biolen implants into a prostate tumor based on the tumor’s size and location. Armed with MRI information and real-time ultrasound imaging, the team uses a needle to place up to 16 implants into the tumor, depending on its configuration in the prostate. The small size of the device allows the researchers to be strategic about how and where to implant it. “We don’t want it to be everywhere in the prostate. We want it to be in the tumor,” says Citrin.
Once inside the patient’s tumor, Biolen releases bicalutamide from a silicone matrix directly into the surrounding tumor tissue. After eight weeks, the team will perform two weeks of localized radiation treatment, and then follow each patient for two years. Biolen implants remain in the prostate long-term, with no subsequent operation anticipated.
“This is highly novel in the setting of prostate cancer,” Citrin says. “I’m hopeful this will be a way to deliver medicine without the side effects but with the same benefit in outcome.” The team will assess the feasibility of the procedure, tumor characteristics that predict response to treatment and the overall quality of life for patients. Citrin hopes the results of this study will support a much larger clinical trial in the future.
If successful, this approach could provide a way to deliver treatment directly into target areas, avoiding negative side effects and harm to healthy tissue. “This could be paradigm-shifting in not only prostate cancer, but in other localized cancers where there’s toxicity to anti-cancer medications,” Citrin says.
Clinicaltrials.gov identifier: NCT04943536
NCI Protocol ID: NCI000328
Official Title: Bicalutamide Implants (Biolen) With Radiation Therapy in Patients With Localized Prostate Cancer (Biolen + RT)
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.