Robert Yarchoan, M.D.
- Center for Cancer Research
- National Cancer Institute
- Building 10, Room 6N106
- Bethesda, MD 20892-1868
- 240-760-6075
- Robert.Yarchoan@nih.gov
RESEARCH SUMMARY
Dr. Yarchoan has played a major role in developing the first successful drugs to treat HIV infection. More recently, his research is focused on the study of and development of treatment for HIV-associated malignancies, with a focus on those caused by Kaposi sarcoma-associated herpesvirus (KSHV). As Chief, Dr. Yarchoan oversees the Branch’s clinical/translational research program, which encompasses the development of novel therapies for HIV infection, the study of the viral causes of HIV-associated malignancies, and the development of novel therapies for HIV-associated malignancies.
Areas of Expertise
Information for Patients
Learn more about our clinical trials and the highly specialized care teams that lead them.

Robert Yarchoan, M.D.
Clinical Trials
Research
The research in the Retroviral Diseases Section includes a laboratory and clinical component, and there is substantial integration and cross-fertilization between these components.
Pathogenesis of Virus-Associated Tumors
One main area of research focus is the study of the pathogenesis of viral-associated tumors, especially those associated with Kaposi's sarcoma-associated herpesvirus (KSHV), also called human herpesvirus-8 (HHV-8). KSHV is the causal agent of Kaposi's sarcoma (KS), primary effusion lymphoma (PEL), and multicentric Castleman disease (MCD). We observed that KS preferentially develops in the feet and other areas of the body with a poor vascular supply. In studying this phenomenon, we discovered that KSHV is induced by hypoxia to undergo lytic replication and to produce factors with direct or indirect angiogenic activity. In further exploring this observation, we found that the genome of KSHV encodes for several hypoxia response elements (HRE) that respond to hypoxia-induced factor (HIF). We are currently exploring the differential upregulation of various KSHV genes by hypoxia and how this may influence the pathogenesis of KSHV-induced tumors. In an extension of this work, we are also exploring the differential upregulation of various cellular genes by two different HIFs, HIF-1 and HIF-2. We are also exploring how LANA interacts with HIF in affecting KSHV and cellular genes, and other factors that can upregulate certain KSHV genes. In particular, we are exploring how spliced X-box protein (XBP) can directly upregulate KSHV viral interleukin-6 (vIL-6); this may play an important role in the pathogenesis of MCD. In addition, we are exploring how KSHV latency-associated nuclear antigen (LANA) is affected by oxidative stress, and in particular its cleavage by cellular caspases.
We are also studying the pathogenesis of KSHV-associated MCD and related disorders. As part of of this effort, we are studying the cytokine dysregulation in MCD and its association with symptomatology. We have also recently identified a new MCD-like syndrome caused by KSHV in which patients have inflammatory symptoms similar to those of MCD, have increase KSHV viral IL-6 levels, but do not have pathological findings of MCD. This syndrome, which we are calling KSHV-associated inflammatory cytokine syndrome (KICS), may account for otherwise unexplained fevers in certain patients with HIV and KSHV co-infection.
Development of Novel Therapies for AIDS-Related Malignancies and HIV Infection
Another main focus of our research is the development of novel therapies for HIV-related malignancies and HIV infection. This work is informed by the pathogenesis-associated work described above. One area of principal interest is Kaposi's sarcoma (KS). Our group is exploring approaches that are based on an understanding of the pathogenesis of this disease but that do not involve the use of cytotoxic chemotherapy. There is evidence that production of virally encoded and cellular angiogenesis-inducing factors by KSHV-infected cells is important in the pathogenesis of KS, and this makes antiangiogenesis approaches attractive to consider. Our group recently showed that bevacizumab (humanized anti-VEGF antibody) has activity in KS, and we are now studying the combination of this with liposomal doxorubicin and also pomalidomide, an analog of thalidomide. We recently initiated protocols to explore targeted therapy for KSHV-MCD and HIV-associated primary central nervous system lymphoma.
Publications
- Bibliography Link
- View Dr. Yarchoan's PubMed Summary
Biography

Robert Yarchoan, M.D.
Dr. Robert Yarchoan received his B.A. from Amherst College with a major in biophysics and his M.D. from the University of Pennsylvania. He trained in internal medicine at the University of Minnesota and Immunology in the Metabolism Branch, NCI. He then joined the laboratory of Dr. Samuel Broder, where he played a major role in the development of the first effective therapies for HIV infection, including zidovudine (AZT), didanosine (ddI), and zalcitabine (ddC). In particular, he led the first clinical trials of these drugs, was a co-inventor of didanosine and zalcitabine as AIDS therapies, and led initial studies of combination anti-HIV therapy. He was a Section Chief in the Medicine Branch from 1991 to 1996 and was named chief of the newly formed HIV and AIDS Malignancy Branch in 1996. Since that time, he has focused much of his research on AIDS-related malignancies, especially tumors caused by Kaposi's sarcoma-associated herpesvirus (KSHV).
Among other honors, he has been awarded the Assistant Secretary for Health Award and the U.S. Public Health Service Outstanding Service Medal, has been inducted as a Fellow of the American Association for the Advancement of Science (AAAS), and is a member of the American Society for Clinical Investigation. In December, 2006, he was a recipient of the first NIH World AIDS Day Award, and in November, 2007, he received the NCI HIV/AIDS Research Excellence Award along with Drs. Samuel Broder, Robert C. Gallo, and Hiroaki Mitsuya. Recent honors and awards include: 2013, an honorary degree (Doctor of Science) from Amherst College; 2014, the Abbott Award in Clinical and Diagnostic Immunology by the American Society for Microbiology; 2016, elected into the Association of American Physicians; 2017, elected into the American Academy of Microbiology.
In December 2007, Dr. Yarchoan also was appointed as the first Director of the NCI Office of HIV and AIDS Malignancy (OHAM). This office, which is in the Office of the Director, NCI, coordinates and prioritizes the HIV/AIDS and AIDS malignancy research portfolio throughout the NCI.
Job Vacancies
We have no open positions in our group at this time, please check back later.
To see all available positions at CCR, take a look at our Careers page. You can also subscribe to receive CCR's latest job and training opportunities in your inbox.
Team
News
Dr. Robert Yarchoan, Chief of HAMB, delivered an opening plenary talk on HIV/AIDS-associated malignancies at the 2023 AACR meeting. He discussed the changes in HIV malignancy as the AIDS epidemic evolved, cancer immunotherapy in people with HIV, and advances in Kaposi sarcoma and related diseases
Lab Life

Going away party for Yi-Quan Wu, who is finishing his post-doc and joining a local Pharma company.

HAMB members give a good send-off to our Post-Bac, Emma Treco, who is off to an MD-PhD Program at the U Wisconsin.

Visit of Coralie Viollet with HAMB lab members July of 2024. Dr. Viollet completed her PhD in the Yarchoan lab several years ago. She is currently a Laboratory Head at AstraZeneca in England and is also on the Board of Directors of the NIH Ox-Cam program.
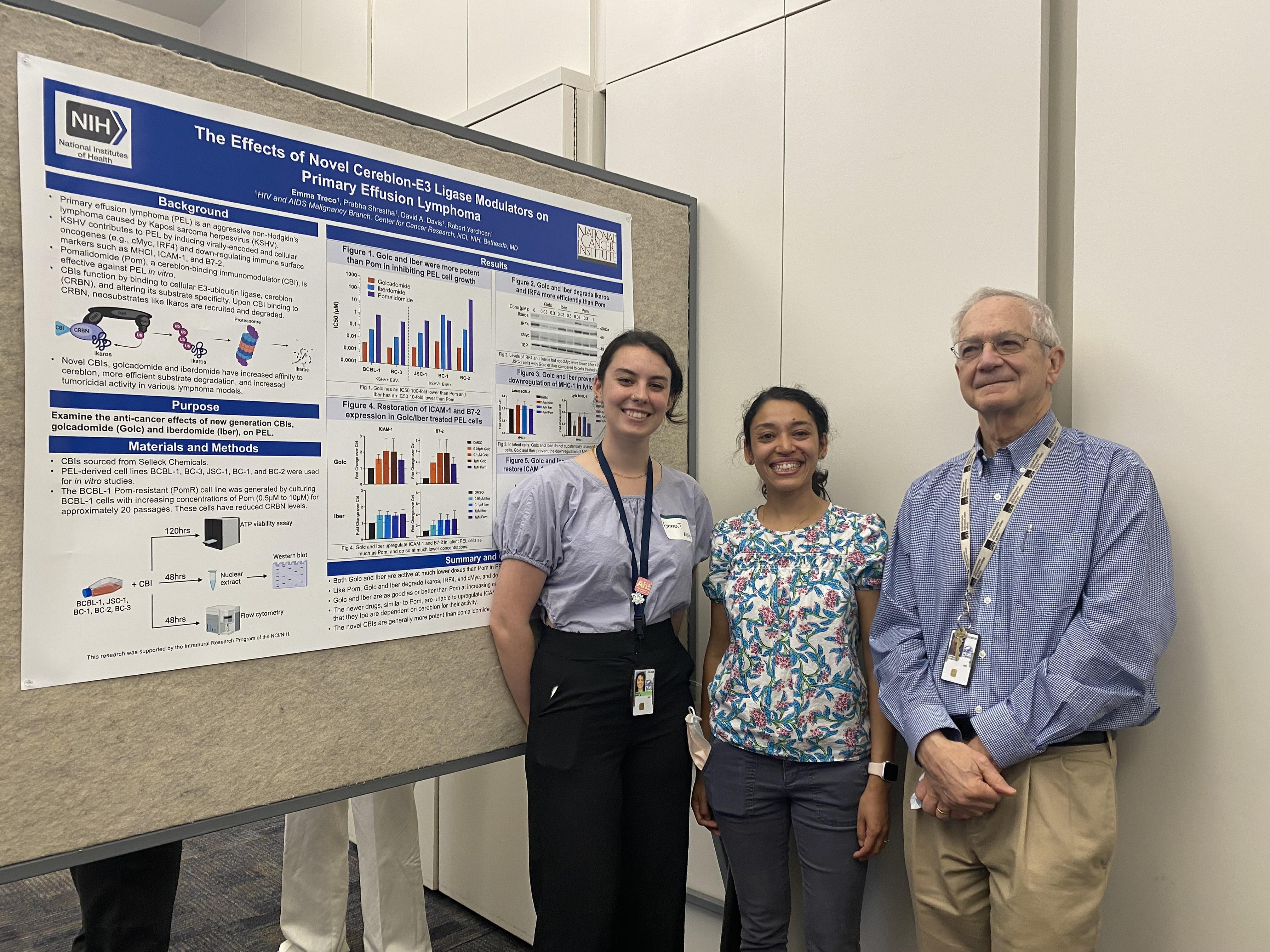
Our postbac, Emma Treco, presenting her poster at the NIH Postbac Poster Day, May 2, 2024. Shown from l to r are Emma Treco, Prabha Shrestha, and Robert Yarchoan.

Members of the Yarchoan lab and others in HAMB got together at a local restaurant in May, 2023, to give a sendoff to Yana Aster, a post-bacc. After 2 years, she is leaving to start an MD/Ph.D. program in the University of Michigan.

A few members of the Yarchoan lab relaxing at the HAMB picnic, September, 2022.

Supper with Dr. Tom Uldrick, Sept. 19, 2019.

Congratulations to Dr. Ramya Ramaswami on her appointment as an Assistant Clinical Investigator (ACI) in HAMB, CCR, NCI! (Celebration August 2019)

Prabha Shrestha (center) receiving a Training Award at the KSHV International Workshop, NYC (July 2, 2019)