Martin J. Schnermann, Ph.D.
- Center for Cancer Research
- National Cancer Institute
- Building 376, Room 225D
- Frederick, MD 21702
- 301-228-4008
- schnermannmj@mail.nih.gov
RESEARCH SUMMARY
The Schnermann lab uses the tools and concepts of modern organic chemistry to discover new molecules for cancer drug delivery and imaging. In the context of drug delivery, we develop innovative chemical strategies to deliver bioactive payloads with high precision. In the area of imaging, we create novel fluorescent molecules with improved properties for in vivo optical imaging and microscopy. While our studies are enabled by core expertise in organic synthesis and molecular design, trainees in the lab are engaged in highly interdisciplinary research using techniques that range from in vitro characterization to in vivo imaging. We embrace a highly collaborative approach with extensive interactions with both intramural and extramural investigators.
Areas of Expertise

Martin J. Schnermann, Ph.D.
Research
New Synthetic Approaches to Small Molecules for Imaging
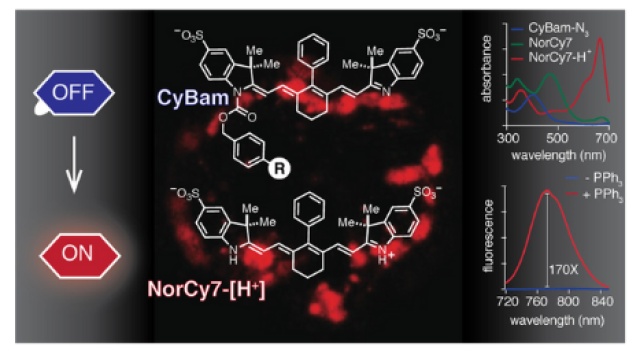
Emerging imaging applications require probes tailored to the specific demands of each method. Our approach is to define the complex reactivity of the cyanine chromophore and apply these chemistries to enable molecular design and probe discovery efforts. Through our now decade-long experience with these molecules, we have significant insight into their preparation, reactivity, and relationships between chemical structure and physical/biological properties. A major component of our efforts is to create novel synthetic methods, which allows us access to previously unknown molecules. We have developed the chemistry to assemble exceptionally bright, polycyclic pentamethine cyanines that are enabling for a variety of advanced microscopy applications. We have also worked extensively to develop novel heptamethine cyanine dyes. These molecules operate in the near-infrared (NIR) range making them applicable to in vivo live animal imaging applications. We developed a highly modified heptamethine cyanine dye, FNIR-tag, that is uniquely resistant to aggregation and provides exceptionally bright bioconjugates. We are also developing bioconjugatable nonamethine indocyanine dyes with emission beyond 1000 nm. These molecules have extended the range of wavelengths available for targeted multicolor in vivo imaging. Finally, we are developing novel turn-on or “fluorogenic” probe chemistries to enable responsive in vivo imaging applications.
Applying Optical Approaches to the Drug Delivery Problem
Recent progress establishing the clinical utility of ADCs has validated the potential of ligand-targeted small molecule delivery strategies. However, the application of these drugs has encountered significant toxicities that limits the scope of their use. Critically, these toxicities rarely result from the mAb binding to its cognate target, but rather from deleterious effects of the hydrophobic small molecule/linker combination on in vivo targeting. Consequently, the chemical components - the linker and payload - require redesign to realize the promise of these strategies. In prior studies, we developed a cyanine-based light-activated ADC strategy. Building on this work and our efforts to optimize mAb conjugates for FGS applications, we are applying optical imaging to define key criteria that determine the tumor targeting of ADCs. These efforts will identify tumor selective protease-cleavable linkers and payloads with improved physical properties. This approach builds on our expertise in optical imaging and probe design, and benefits from CCR collaborations with experts in mAb and therapeutic development.
Publications
Harnessing Cyanine Reactivity for Optical Imaging and Drug Delivery
Norcyanine-Carbamates Are Versatile Near-Infrared Fluorogenic Probes
In vivo activation of duocarmycin–antibody conjugates by near-infrared light
Cyanine conformational restraint in the far-red range
Biography

Martin J. Schnermann, Ph.D.
Dr. Schnermann attended Colby College and graduated in 2002 with degrees in Chemistry and Physics. At Colby, he worked with Prof. Dasan Thamattoor in the areas of physical organic chemistry and photochemistry. After a year at Pfizer Research and Development (Groton, CT) as an associate in the medicinal chemistry division, he moved to the Scripps Research Institute. During his graduate studies, he performed research on the total synthesis and biological evaluation of anticancer natural products with Prof. Dale Boger and obtained a Ph.D. in 2008. He then completed an NIH-postdoctoral fellowship with Prof. Larry Overman at the University of California, Irvine. At Irvine, he developed light-mediated reactions to enable the synthesis of complex natural products. In addition, working with Prof. Christine Suetterlin, he pursued chemical biology and imaging studies of organelle specific probes. In 2012, Dr. Schnermann joined the Chemical Biology Laboratory at the National Cancer Institute, where his research focuses on the synthesis and development of new small-molecule imaging agents for cancer treatment and diagnosis. In 2018, Dr. Schnermann was promoted to Senior Investigator.
Job Vacancies
We have no open positions in our group at this time, please check back later.
To see all available positions at CCR, take a look at our Careers page. You can also subscribe to receive CCR's latest job and training opportunities in your inbox.
Team
News
A funded postdoctoral position is available within the Chemical Biology Laboratory at the National Cancer Institute, Frederick, Maryland. Research projects center on the development of new approaches for cancer imaging and drug delivery. The lab is focused on the synthesis and development of small molecules, with a specific interest on methods that function in complex organisms. The NCI is an excellent environment for postdoctoral study. Postdoctoral scholars will have access to first-rate scientific resources, strong training opportunities, and a close community of researchers working on critical problems that link chemistry to cancer biology. Prospective candidates should have a strong background in synthetic organic chemistry, photochemistry, and/or chemical biology and be motivated to actively contribute to interdisciplinary projects. Potential applicants should contact Dr. Martin Schnermann (martin.schnermann@nih.gov) to express interest (please enclose a C.V. and a letter of support).
Covers

Core remodeling leads to long wavelength fluoro-coumarins
Low molecular weight, uncharged far-red and NIR dyes would be enabling for a range of imaging applications. Rational redesign of the coumarin scaffold leads to Fluoro-Coumarins (FCs), the lowest molecular weight dyes with emission maxima beyond 700, 800, and 900 nm. FCs display large Stokes shifts and high environmental sensitivity, with a 40-fold increase in emission intensity in hydrophobic solvents. Untargeted variants exhibit selective lipid droplet and nuclear staining in live cells. Furthermore, sulfo-lipid derivatization enables active targeting to the plasma membrane. Overall, these studies report a promising platform for the development of biocompatible, context-responsive imaging agents.
See: Core remodeling leads to long wavelength fluoro-coumarins by Siddharth S. Matikonda, Joseph Ivanic, Miguel Gomez, Gabrielle Hammersley and Martin J. Schnermann in Chemical Science, 2020, 11, 7302-7307.

Cyanine polyene reactivity: scope and biomedical applications
Cyanines are indispensable fluorophores that form the chemical basis of many fluorescence-based applications. A feature that distinguishes cyanines from other common fluorophores is an exposed polyene linker that is both crucial to absorption and emission and subject to covalent reactions that dramatically alter these optical properties. Over the past decade, reactions involving the cyanine polyene have been used as foundational elements for a range of biomedical techniques. These include the optical sensing of biological analytes, super-resolution imaging, and near-IR light-initiated uncaging. This review surveys the chemical reactivity of the cyanine polyene and the biomedical methods enabled by these reactions. The overarching goal is to highlight the multifaceted nature of cyanine chemistry and biology, as well as to point out the key role of reactivity-based insights in this promising area.
See: Cyanine polyene reactivity: scope and biomedical applications by Alexander P. Gorka, Roger R. Nani and Martin J. Schnermann in Organic & Biomolecular Chemistry, 2015, 13, 7584-7598.