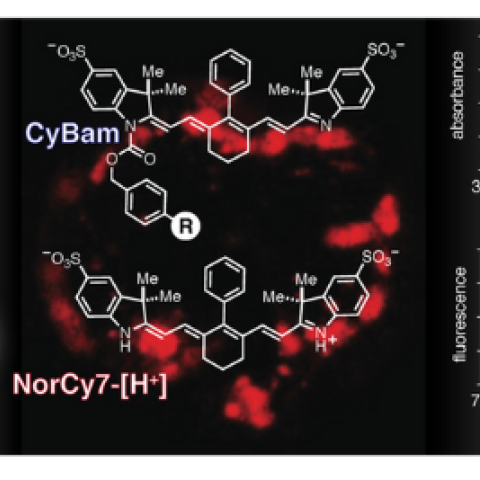
Near-infrared compounds for visualizing enzymatic activity in living organisms
Image Credit: Usama SM, et al. J Am Chem Soc. 2021 Apr 12.
Newly developed chemical compounds are safe and highly effective in fluorescently highlighting microscopic metastases, according to research findings from CCR scientists and clinicians. These new compounds, called cyanine carbamates, could have great utility in complex living systems based on experiments done in mouse models of metastatic ovarian cancer. This study appeared April 12, 2021, in the Journal of the American Chemical Society.
“When we look at the night sky, we can see stars much better than in the daytime. Similarly, we can detect tumors with a higher degree of sensitivity if cancer cells are fluorescing against a minimally cluttered backdrop because they stand out much more clearly, and that’s what our new compounds are designed to do,” says Hisataka Kobayashi, M.D., Ph.D., Senior Investigator in the Molecular Imaging Branch. “In the clinic, ovarian cancer frequently micro-metastasizes and implants small tumors in the nearby peritoneum. While surgeons try to remove as many metastases as possible, optimally visualizing small cancer nodules with fluorescent probes like ours could ensure that all, or almost all, of the cancer metastases are removed.”
Understanding which compounds emit the best fluorescent signal is important. Visible wavelengths are absorbed by biomolecules, especially hemoglobin, and are good for seeing cells and tissue slices under a microscope. But near-infrared wavelength-emitting compounds allow scientists to see deeper into tumors than is possible with visible wavelengths.
“Figuring out what molecules can both be activated and emit long wavelength light is a tricky problem, and that is part of the reason we are excited about the compounds we designed,” says Martin J. Schnermann, Ph.D., Senior Investigator in the Chemical Biology Laboratory and corresponding author of the study. “Because the tissues in our body auto-fluorescence at low levels in the near-infrared range, using these compounds minimizes background signals and allows us to see the fluorescent signal more easily.”
Another positive aspect of these new compounds is their low toxicity. Some molecules and nanoparticles designed for use with infrared wavelengths can accumulate in healthy tissue. Because these new compounds do not do this, they are efficiently eliminated from the body and have been proven to be nontoxic. Also, the high sensitivity of the compounds allows for low dosages, which further minimizes toxicity.
“We are continuing to improve the chemistry of these compounds and think there is significant potential for these molecules to soon be applied in a clinical setting. There are also other exciting applications in preclinical settings, such as using these molecules to develop and test drug delivery approaches in animals,” Schnermann says.
The researchers note that they hope to use these compounds in fresh surgical specimens to see if any residual cancer exists. They also believe the compounds will aid in minimizing the amount of time for surgical resection and hence limit impacts on patients.