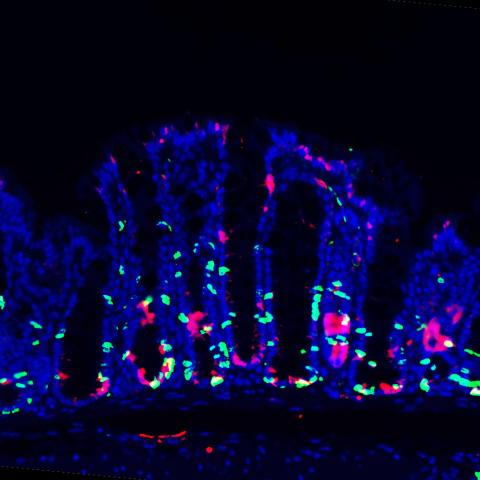
An image of mouse colon tissue (blue), proliferating stem cells (green) and PEDF (red). Image credit: Girak Kim, et al.
Liver signaling has been found to play an important role in directing intestinal stem cell repair of injured gut tissue through a protein called PEDF. The finding, by CCR researchers, was published in Cell on January 26, 2024, and highlights the influence of organ-to-organ communication in human health and disease.
For decades, scientists have understood that the liver and gut exchange fluids and signaling molecules — a system called the gut-liver axis — and diseases of these organs often co-occur. However, the mechanisms by which the gut-liver axis regulates the health of the intestines have remained largely unknown.
CCR researchers, led by Chuan Wu, M.D., Ph.D., Stadtman Investigator in the Experimental Immunology Branch, conducted a series of experiments in healthy mice and those with gut disease. They found that the PEDF protein, which is continuously made by the liver, normally binds to and stops intestinal stem cells from being able to divide and grow — until the gut is impaired.
These stem cells, Wu explained, reside at the base of tiny intestinal folds and expand when the intestine is harmed by microbes or other damaging substances. This must be tightly regulated, he warned, “because if they divide too much, cancer can develop.”
Through additional experiments in mice, Wu’s team discovered that the intestines, when damaged, send a potent signal to the liver that specifically dials down the liver’s PEDF production via another protein called PPAR-alpha. This, in turn, releases the hold on stem cell expansion, allowing them to grow and heal the inflamed tissue.
Consistent with what the researchers saw in mice, they found that tissue from humans with inflammatory bowel disease also had a reduced amount of PEDF, presumably a consequence of the need for intestinal stem cell activity.
Unexpectedly, the researchers stumbled upon a surprising revelation. For years, doctors have reported increased cases of colitis, a type of gut disease, in patients given a drug called fenofibrate to treat high cholesterol by lowering lipid counts. Fenofibrate enhances PPAR-alpha, which — as shown in the study — subsequently sustains PEDF and halts stem cell repair of diseased tissue. For the first time, the researchers showed that this unfortunate side effect of fenofibrate could be eliminated by neutralizing the PEDF protein in mice.
Wu’s hope is that this study helps doctors appreciate that organ-to-organ communication can be a new therapy strategy. Wu also notes: “Stem cells are everywhere,” lending hope to the possibility that PEDF manipulation could be used to target stem cells beyond the gut and elsewhere in the body.