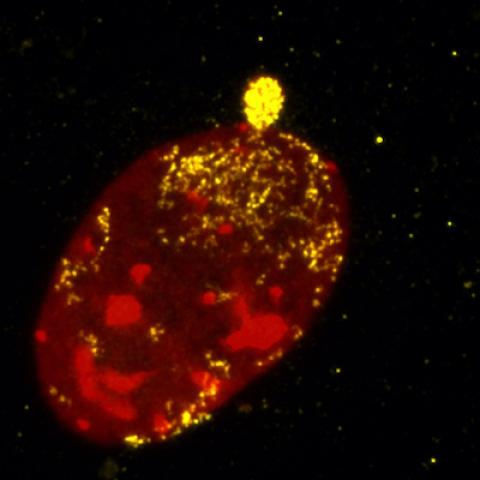
Genomic amplification of the oncogene c-myc (yellow) in a mouse model of human colorectal cancer.
Image credit: Thomas Ried, Center for Cancer Research
Adults with metastatic, recurrent or refractory small bowel and colorectal cancers may be eligible to participate in a new clinical trial at the NIH Clinical Center.
Julius Strauss, M.D., Assistant Research Physician in the Laboratory of Tumor Immunology and Biology, is leading a clinical trial evaluating a combination of immunotherapy drugs to see if they can shrink advanced tumors of the small bowel, colon and rectum. When small bowel/colorectal cancers spread to other parts of the body, return after treatment, or stop responding to treatment, they cannot be cured. Participants will be put into study groups that will each receive a different combination of three, or possibly four drugs.
CV301 targets certain proteins expressed by tumors. M782 blocks pathways that cancer cells use to avoid attack by the immune system. N-803 improves the immune system’s ability to kill cancer cells. Some participants will get NHS-IL12 which triggers the immune system to fight tumors. Researchers want to see if this combination is a better treatment for these cancers.
Clinicaltrials.gov identifier: NCT04491955
NCI Protocol ID: NCI-20-C-0138
Official Title: Phase II Trial of Combination Immunotherapy in Subjects With Advanced Small Bowel and Colorectal Cancers
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.