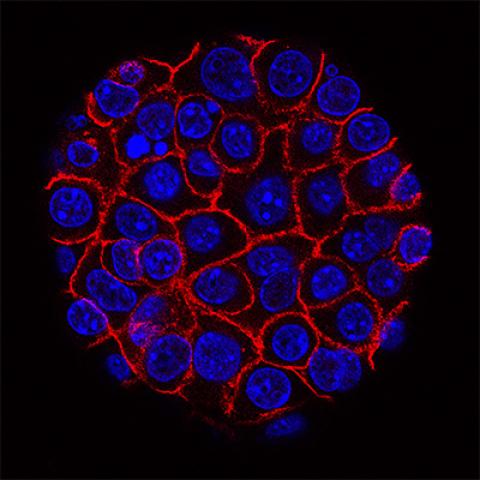
This image shows pancreatic cancer cells (nuclei in blue) growing as a sphere encased in membranes (red).
Photo courtesy of NCI Visuals Online
A clinical phase II trial at the NIH Clinical Center will test the effectiveness of a monoclonal antibody used immediately before and after surgery for patients with primary pancreatic and bile duct cancer and those who have liver metastasis from colorectal cancer. The trial is investigating whether this treatment can prevent the disease from coming back after surgery.
Gastrointestinal tumors—primary pancreatic and bile duct (chorlangiocarimona) cancer and liver metastasis from colorectal cancer—are very aggressive, with high one-year recurrence rates and marginal five-year survival rates. According to NCI SEER data, the one-year survival rate for pancreatic cancer is about 20 percent, and the five-year survival rate is 7 percent. The statistics for bile duct cancer are equally sobering with a five-year survival rate of about 18 percent.
This new clinical trial at the NIH Clinical Center aims to address this problem head-on with a focus on high recurrence rates.
“We know that between 40 and 50 percent of patients with these aggressive tumors have recurrences, often within the first year,” said Jonathan Hernandez, M.D., Investigator in CCR’s Surgical Oncology Program, and the lead investigator for the trial. “We have a good idea why this is the case. From the time the tumor begins to grow in an organ, cells that are shed from the tumor can travel throughout the body. While most of these cells die or remain dormant, some begin to grow, resulting in a life-threatening recurrence of the cancer.”
Operable Gastrointestinal Tumors: Clinical Trial Conversation
Jonathan Hernandez, M.D., Surgical Oncologist at the Center for Cancer Research in the National Cancer Institute, discusses a phase II clinical trial for patients with operable gastrointestinal tumors.
Chemotherapy cannot be given for about two months following surgery, largely because the body needs that time to heal. But during that interval, cancer cells throughout the body have had an opportunity to grow. “The microtumors thrive in the inflammatory conditions created as a result of the surgery, which makes patients particularly vulnerable during this period,” added Hernandez.
The trial’s goal is to bridge this gap period by giving patients the monoclonal antibody MVT-5873, which targets an antigen called CA 19-9 produced by the tumor cells. This antibody isn’t targeting the tumor that’s going to be surgically removed. Instead, it’s going after micrometastases that cannot be seen yet. The antibody works by binding to individual tumor cells and causes them to die.
Patients will be given the antibody before surgery and then immediately following it, during the critical 2-month period when the body is healing. Researchers will be testing how safe the treatment is before and after surgery and determine the highest doses that can be tolerated. When patients are switched over to standard adjuvant chemotherapy, they will be taken off the trial protocol but will continue to be followed. The trial will include between 65 and 90 patients.
The trial builds on an earlier phase I trial sponsored by MabVax Therapeutics, which developed the antibody, and was led by Eileen O'Reilly, M.D., associate director of the David M. Rubenstein Center for Pancreatic Cancer Research at Memorial Sloan Kettering Cancer Center. The six patients in the study were given MVT-5873 along with standard-of-care chemotherapy. All the patients had measurable reductions in tumor volume and tolerated the treatment well.
As a result of these positive findings, the MabVax team is conducting a larger, multicenter trial. They are currently recruiting as many as 68 patients. The new trial will look at patients receiving the monoclonal antibody as well as those receiving chemotherapy.
The two trials should provide meaningful data about the effectiveness of this treatment. “Our hope is that we can cut down on early recurrences, which typically take place within the first year after surgery,” said Hernandez. “That gives us a readily measurable endpoint for the study. If we’re successful, down the road this treatment may become part of standard care.”
To learn more about this trial, visit our website.
Clinicaltrials.gov identifier: NCT03801915
Title: Perioperative MVT-5873, a Fully Human Monoclonal Antibody Against a CA 19-9 Epitope, for Operable CA 19-9 Producing Pancreatic Cancers, Cholangiocarcinomas, and Metastatic Colorectal Cancers
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.