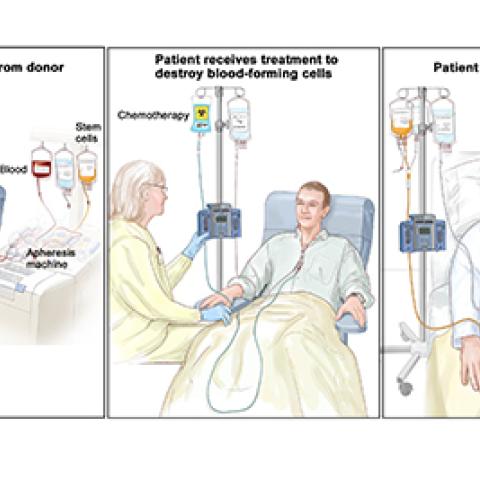
Stem cell transplant process
Photo courtesy of NCI Visuals Online
Patients with relapsed or refractory peripheral T-cell lymphoma (PTCL) may be eligible to participate in a new clinical trial at the NIH Clinical Center.
Jennifer A. Kanakry, M.D., of the Experimental Transplantation and Immunology Branch, is leading a clinical trial for patients with relapsed or refractory peripheral PTCL. This type of lymphoma is a rare, often fast-growing cancer of white blood cells (T cells), which help the immune system fight infection. With PTCL, some T cells start to grow out of control and accumulate in the body. PTCL is usually treated with a combination of chemotherapies, but if the disease returns after treatment or doesn’t respond to treatment, then a blood stem cell transplant from a healthy donor is often recommended to use the immune system to fight the lymphoma. Blood stem cell transplant can cure some people with PTCL, but there can be serious complications and side effects. Investigators in this trial want to see how a new approach to blood stem cell transplant impacts patients with PTCL while also decreasing the side effects.
Clinicaltrials.gov identifier: NCT03922724
NCI Protocol ID: NCI-19-C-0085
Official Title: Phase II Trial of Allogeneic Hematopoietic Cell Transplantation for Peripheral T Cell Lymphoma
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.