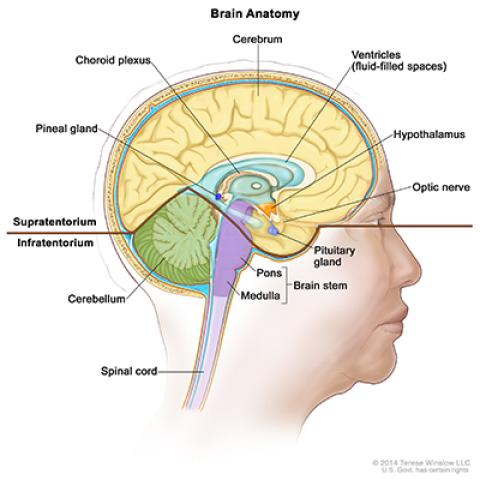
Drawing of the inside of the brain showing the supratentorium (the upper part of the brain) and the infratentorium (the lower back part of the brain).
Photo credit: NCI Visuals Online
Patients with recurrent low-grade and anaplastic ependymomas of the brain and spinal cord may be eligible to participate in a new clinical trial at the NIH Clinical Center.
Ependymomas are rare tumors that arise in the ependyma, the thin membrane that lines fluid-filled cavities in the brain and spinal cord. Anaplastic refers to cancer cells that are dividing rapidly and have little or no resemblance to normal cells. There are limited treatment options for ependymomas that have already been treated with standard therapies such as surgery and radiation. Mark Gilbert, M.D., Chief of the Neuro-Oncology Branch, is leading a trial of marizomib, a drug that targets ependymomas that have a specific genetic signature by slowing the spread of cancer cells. Marizomib may disrupt cellular processes within these specific tumor cells, leading to cell death. Investigators want to see if marizomib can lengthen the time it takes for recurrent ependymomas to get worse.
Clinicaltrials.gov identifier: NCT03727841
NCI Protocol ID: NCI-19-C-0011
Official Title: Phase II Clinical Trial of Marizomib for Recurrent Low-Grade and Anaplastic Supratentorial, Infratentorial and Spinal Cord Ependymoma
The Center for Cancer Research is NCI’s internal cancer center, a publicly funded organization working to improve the lives of cancer patients by solving important, challenging and neglected problems in cancer research and patient care. Highly trained physician-scientists develop and carry out clinical trials to create the medicines of tomorrow treating patients at the world’s largest dedicated research hospital on the campus of the National Institutes of Health in Bethesda, Maryland.
For more information on CCR clinical trials click here, and subscribe to have the latest CCR clinical trials sent directly to your inbox.