Raffit Hassan, M.D.
- Center for Cancer Research
- National Cancer Institute
- Bldg 10; 10 Center Drive room: 4E-5330
- Bethesda, MD 20892
- 240-760-6232
- hassanr@mail.nih.gov
RESEARCH SUMMARY
Dr. Hassan has played a pivotal role in validating the tumor differentiation antigen mesothelin as a target for cancer therapy and development of mesothelin targeted immunotherapy. His current research focus is on developing mesothelin directed adoptive cellular therapies including clinical trials of T cell receptor fusion constructs, gavo-cel and TC-510. Additionally, his lab has developed highly effective anti-mesothelin hYP218 CAR T cells that bind mesothelin close to the cell membrane and will enter the clinic later this year. The Hassan lab also studies the genetics of mesothelioma and is leading a prospective trial of early cancer detection in patients and their family members with germline mutations in the BAP1 cancer pre-disposition gene.
Areas of Expertise
Information for Patients
Learn more about our clinical trials and the highly specialized care teams that lead them.
Raffit Hassan, M.D.
Clinical Trials
Research
Dr. Hassan's clinical and laboratory research is focused on developing mesothelin targeted immunotherapy to treat patients with malignant mesothelioma and other solid tumors. While working as a postdoctoral fellow in the laboratory of Dr. Ira Pastan, Dr. Hassan did the initial preclinical studies to exploit mesothelin as a target for cancer therapy and has played a key role in bringing mesothelin-directed therapies to the clinic. These include anti-mesothelin immunotoxins (SS1P and LMB-100), chimeric monoclonal antibody to mesothelin (Amatuximab) and anti-mesothelin antibody drug conjugate (BAY 94-9343).
His lab has recently developed highly active CAR T cells (hYP218) that target a membrane proximal epitope of mesothelin and clinical trial of hYP218 CAR T cells to treat patients with treatment refractory solid tumors is expected to open soon at the NCI. His ongoing laboratory studies are focused on making CAR T cells effective to treat solid tumors by overcoming tumor immune suppressive factors; improving persistence of CAR T cells and using mesothelin directed NK cells for cellular therapy.
In addition, he is interested in mesothelioma genetics and has initiated a long-term prospective trial of cancer screening in patients and their asymptomatic family members harboring pathogenic mutations in the BAP1 gene for early detection of different cancers to which they are predisposed. This protocol will provide definitive answers about incidence of different cancers in these families and whether early cancer detection will improve their outcomes.
Publications
Biography
Raffit Hassan, M.D.
Dr. Raffit Hassan is a medical oncologist and senior investigator at NCI. He received his medical degree from the University of Kashmir, India in 1988 and did his internship and residency training in internal medicine at Sisters Hospital, University of Buffalo and subsequently completed his medical oncology fellowship at the NCI. After finishing his fellowship training he joined the medical faculty at the University of Oklahoma in 1998. He returned to NCI as a tenure track investigator in 2002 and received tenure in 2008. Dr. Hassan is a recipient of the ASCO Career Development Award, the NIH Patient Oriented Research Career Development Award and the Pioneer Award from the Mesothelioma Foundation.
Job Vacancies
We have no open positions in our group at this time, please check back later.
To see all available positions at CCR, take a look at our Careers page. You can also subscribe to receive CCR's latest job and training opportunities in your inbox.
Team
News
Covers
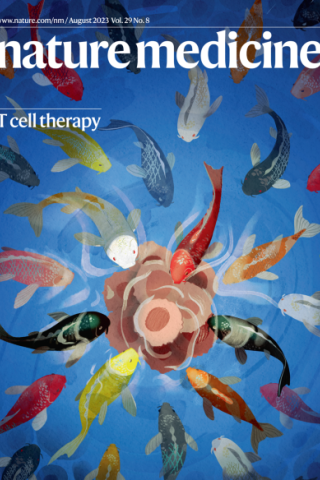
Mesothelin-targeting T cell receptor fusion construct cell therapy in refractory solid tumors: phase 1/2 trial interim results.
T cell therapy
In this issue, Hassan et al. show that engineered HLA-independent T cells specific for mesothelin, an antigen that is overexpressed in many cancers, exhibit toxicities as well as anti-tumor activity in patients with treatment-refractory mesothelioma or ovarian cancer. The school of omnivorous koi fish on the cover represents the T cells, which are shown attacking a solid tumor.
Image: Erina He, NIH Medical Arts. Cover design: Marina Spence
R. Hassan, M. Butler, R. E. O'Cearbhaill, D. Y. Oh, M. Johnson, K. Zikaras, M. Smalley, M. Ross, J. L. Tanyi, A. Ghafoor, N. N. Shah, B. Saboury, L. Cao, A. Quintás-Cardama, D. Hong, Mesothelin-targeting T cell receptor fusion construct cell therapy in refractory solid tumors: phase 1/2 trial interim results. Nat Med 29, 2099-2109 (2023).

Enhanced efficacy of mesothelin-targeted immunotoxin LMB-100 and anti-PD-1 antibody in patients with mesothelioma and mouse tumor models
Improving Immunotherapy: A mesothelin-targeted immunotoxin enhances the anti-tumor activity of immune checkpoint inhibitor in patients with mesothelioma and in mice.
Q. Jiang, A. Ghafoor, I. Mian, D. Rathkey, A. Thomas, C. Alewine, M. Sengupta, M. A. Ahlman, J. Zhang, B. Morrow, S. M. Steinberg, I. Pastan, R. Hassan, Enhanced efficacy of mesothelin-targeted immunotoxin LMB-100 and anti-PD-1 antibody in patients with mesothelioma and mouse tumor models. Sci Transl Med 12, (550):eaaz7252 (2020).

Clinical Response of Live-Attenuated, Listeria monocytogenes Expressing Mesothelin (CRS-207) with Chemotherapy in Patients with Malignant Pleural Mesothelioma
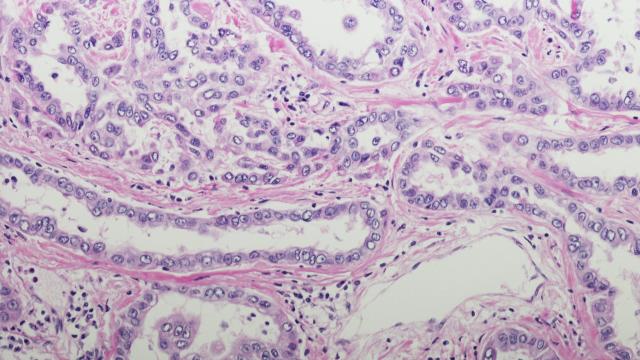
The cover shows a section of a tumor biopsy from a patient with malignant mesothelioma. Multiplex immunohistochemical staining reveals different immune cell populations infiltrating the tumor.
R. Hassan, E. Alley, H. Kindler, S. Antonia, T. Jahan, S. Honarmand, N. Nair, C. C. Whiting, A. Enstrom, E. Lemmens, T. Tsujikawa, S. Kumar, G. Choe, A. Thomas, K. McDougall, A. L. Murphy, E. Jaffee, L. M. Coussens, D. G. Brockstedt, Clinical Response of Live-Attenuated, Listeria monocytogenes Expressing Mesothelin (CRS-207) with Chemotherapy in Patients with Malignant Pleural Mesothelioma. Clin Cancer Res 25, 5787-5798 (2019).

Anaplastic Lymphoma Kinase Gene Rearrangement in Children and Young Adults With Mesothelioma
Peritoneal mesothelioma with ALK-STRN fusion mapping breakpoint to exon 3 of STRN and intron 19 of ALK. The resulting fusion protein contains domains for the Striatin protein and the kinase domain for ALK.
I. Mian, Z. Abdullaev, B. Morrow, R. N. Kaplan, S. Gao, M. Miettinen, D. S. Schrump, V. Zgonc, J. S. Wei, J. Khan, S. Pack, R. Hassan, Anaplastic Lymphoma Kinase Gene Rearrangement in Children and Young Adults With Mesothelioma. J Thorac Oncol 15, 457-461 (2020).